
Heat in an oven to a low controlled temperature to remove gases or to harden a binder.Īmount or quantity of core or mold sand or other material prepared at one time. In steel, a solid solution in which gamma iron is the solvent.Īny steel containing sufficient alloy to produce a stable austenitic (gamma iron) crystalline structure at ambient temperatures. The face-centered-cubic phase of iron and steel, also referred to as gamma iron. Similarly, as drawn, as forged, and as rolled.Īmerican Society for Nondestructive Testing.Īmerican Society for Testing and Materials. Referring to metal which has not received finishing (beyond gate removal or sandblasting) or treatment of any kind including heat treatment after casting. The AOD process improves metal cleanliness and thus gives superior mechanical properties.Īn aging treatment above room temperature. Heating to and holding at a suitable temperature, followed by cooling at a suitable rate to lower the hardness or alter other mechanical or physical properties.Ĭompounds applied to metallic surfaces to prevent surface carbonization.Ī secondary refining process in which argon, oxygen and nitrogen are injected into a molten bath of steel. Steel containing significant quantities of alloying elements other than carbon and the commonly accepted amounts of manganese, silicon, sulfur, and phosphorus.īody-centered cubic type of pure iron stable below 1670☏ (910☌).Ī form or stage of martensite of somewhat arbitrary distinction, probably representing the least developed and most distorted stage in the transformation of austenite to martensite at ordinary temperatures. Usually possesses qualities different from those of the components. In a foundry, the clearance specified difference in limiting sizes, as minimum clearance or maximum interference between mating parts, as computed arithmetically.Ī substance having metallic properties and composed of two or more chemical elements of which at least one is metal. Scale left on ferrous metal in processing, usually from heating in presence of air.  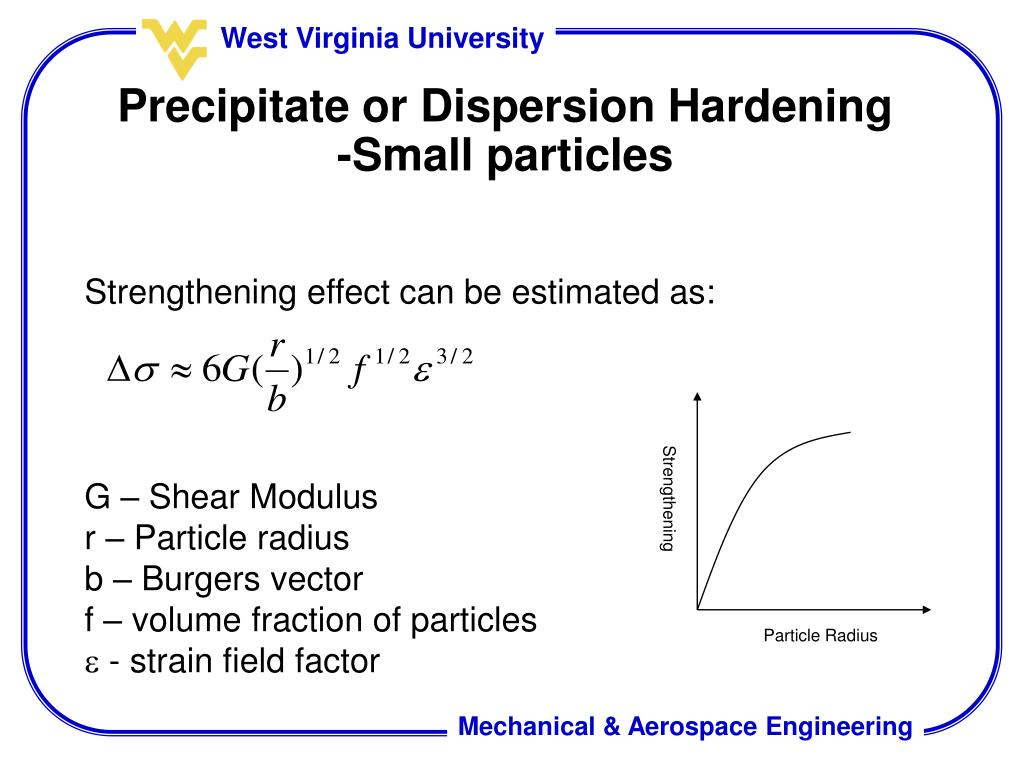
The change in properties is often, but not always, due to a phase change (precipitation), but never involves a change in chemical composition of the metal or alloy.Īccelerated cooling of alloy in an air stream from temperatures above the Ac3 temperature. Hardening by aging, usually after rapid cooling or cold working.Ī change in properties of metals and alloys which occurs slowly at room temperature and will proceed rapidly at higher temperatures. Additionally, it is used on alloys like copper that can be used for roofing, building ships and many other applications.The displacement and/or detachment of metallic particles from a surface as a consequence of being exposed to flowing solids, fluids or gases.ĭegree of resistance of a material to abrasion or wear.Įmbrittlement during pickling due to absorption of hydrogen. Precipitation hardening is commonly used for fabricating parts that must withstand extremely high temperatures, such as turbo chargers and pressure vessels. These prevent dislocations, making it difficult for lose particles to cut through the material. Impurities that are built via the precipitation or hardening technique can cause disruption to the crystal lattice structure in aluminum, stainless steel and other types of alloys. However, this consumes more time-from days to weeks. Another way to achieve precipitate hardening is through natural aging. Excessive soaking or temperatures can result in reduced precipitates, which can promote high ductility and less strength. Precision is essential when calculating the time and temperature. The time for soaking combined with the aging temperature is identified by the intended strength required for the product. Afterward, the solution undergoes soaking for two to 20 hours. This can be accomplished by subjecting the solution to elevated temperatures. Once the quenching phase has been completed and the solution has undergone aging, artificial aging takes place. It is a vital step in strengthening solution, as it prepares the material for the succeeding step of hardening precipitation. This can be accomplished in air or water, or the mixture of these two. The solution’s high saturation, or supersaturation, is obtained by quenching. The mixture produced can be highly saturated and can be readily subjected to treatment. This process can last for as short as an hour to a maximum of 20 hours before the precipitate is completely dissolved. The process of precipitating solid materials begins with heating to extremely elevated temperatures to achieve dissolution. Corrosionpedia Explains Precipitation Hardening
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
